HCM Medication
About Mavacamten
(Camzyos™) & Aficamten
(MYQORZO™)
Mavacamten™ is a first-in-class, myosin inhibitor for the treatment of patients with symptomatic obstructive hypertrophic cardiomyopathy. It is produced by Bristol Myers Squibb.
Aficamten™ is a second-in-class, myosin inhibitor for the treatment of patients with symptomatic obstructive hypertrophic cardiomyopathy developed and produced by Cytokinetics.
Cardiac Myosin Inhibitors (CMIs) treat HCM at its source rather than managing symptoms. Research is currently underway for uses with other types of heart disease, including non-obstructive HCM and heart failure with preserved ejection fraction (HFpEF).
CMIs may reduce the number of people who need to go on to have septal reduction therapy (SRT), an invasive procedure, which is a standard therapy for severe HCM.
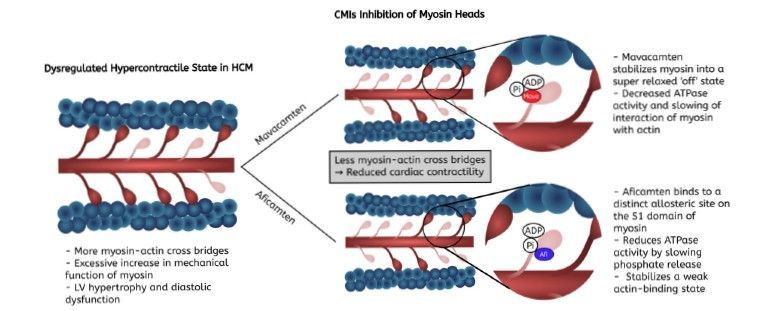
"Mechanisms of action of mavacamten and aficamten, showing their differing binding sites on the cardiac myosin in hypertrophic cardiomyopathy. The figure illustrates how mavacamten (“Mava” in bright red) and aficamten (“Afi” in bright blue) change the activity of myosin heads. Mavacamten binds to catalytic domains of the myosin S1 head, stabilizing an “off” state by decreasing available myosin heads for actin binding. Aficamten binds to a distinct allosteric site from mavacamten, specifically slowing phosphate release in return reducing crossbridge binding. Both agents through differing mechanisms reduce cardiac hypercontractility in hypertrophic cardiomyopathy. Abbreviations: HCM: hypertrophic cardiomyopathy; LV: left ventricle."
https://www.researchgate.net/figure/Mechanisms-of-action-of-mavacamten-and-aficamten-showing-their-differing-binding-sites_fig2_393345936
Bristol Myers Squibb
Camzyos™ Links
Online Information click here: CAMZYOS PATIENT PORTAL
Camzyos - Telephone support line for HCMA Members: 888-694-2059
The information line can provide answers to questions about access and the specific programs available through the MyCamzyos program.
CAMZYOS Medication Guide
CAMZYOS U.S. Prescribing Information
CAMZYOS REMS Program
To determine eligibility and apply for financial assistance, visit
HealthWell’s Cardiomyopathy – Medicare Access Fund page.
Cytokinetics
MYQORZO™ Links
MYQORZO & You - personalized support with a MYQORZO Navigator
MYQORZO - Telephone support : 833-MYQORZO (833-697-6796)
The information line can provide answers to questions about access and the specific programs available through the MYQORZO & You program.
MYQORZO Medication Guide
MYQORZO Brochure
MYQORZO Getting Started
MYQORZO REMS Guide
MYQORZO & You: Patient Access & Affordability
Bighearted Warrior Tour Camzyos™ team at Bristol Myers Squibb
Bighearted Warrior Tour
MYQORZO™ team at Cytokinetics
- Rader F, Choudhury L, Saberi S, et al. Updated cumulative results of treatment with mavacamten from the EXPLORER-LTE cohort of the MAVA-LTE study in patients with obstructive hypertrophic cardiomyopathy. Presented at: ACC 2022. April 3, 2022. Washington, DC.
- Hegde SM, Lester SJ, Solomon SD, Michels M, Elliott PM, Nagueh SF, Choudhury L, Zemanek D, Zwas DR, Jacoby D, Wang A, Ho CY, Li W, Sehnert AJ, Olivotto I, Abraham TP. Effect of Mavacamten on Echocardiographic Features in Symptomatic Patients With Obstructive Hypertrophic Cardiomyopathy. J Am Coll Cardiol. 2021 Dec 21;78(25):2518-2532. doi: 10.1016/j.jacc.2021.09.1381.
- Xie J, Wang Y, Xu Y, Fine JT, Lam J, Garrison LP. Assessing health-related quality-of-life in patients with symptomatic obstructive hypertrophic cardiomyopathy: EQ-5D-based utilities in the EXPLORER-HCM trial.
- J Med Econ. 2022 Jan-Dec;25(1):51-58. doi: 10.1080/13696998.2021.2011301.
Burstein Waldman C, Owens A. A plain-language summary of the EXPLORER-HCM study: mavacamten for obstructive hypertrophic cardiomyopathy. Future Cardiol. 2021 Oct;17(7):1269-1275. doi: 10.2217/fca-2021-0044. Epub 2021 May 21. - Spertus JA, Fine JT, Elliott P, Ho CY, Olivotto I, Saberi S, Li W, Dolan C, Reaney M, Sehnert AJ, Jacoby D. Mavacamten for treatment of symptomatic obstructive hypertrophic cardiomyopathy (EXPLORER-HCM): health status analysis of a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2021 Jun 26;397(10293):2467-2475. doi: 10.1016/S0140-6736(21)00763-7. Epub 2021 May 15.
- Olivotto I, Oreziak A, Barriales-Villa R, Abraham TP, Masri A, Garcia-Pavia P, Saberi S, Lakdawala NK, Wheeler MT, Owens A, Kubanek M, Wojakowski W, Jensen MK, Gimeno-Blanes J, Afshar K, Myers J, Hegde SM, Solomon SD, Sehnert AJ, Zhang D, Li W, Bhattacharya M, Edelberg JM, Waldman CB, Lester SJ, Wang A, Ho CY, Jacoby D; EXPLORER-HCM study investigators. Mavacamten for treatment of symptomatic obstructive hypertrophic cardiomyopathy (EXPLORER-HCM): a randomized, double-blind, placebo-controlled, phase 3 trial. Lancet. 2020 Sep 12;396(10253):759-769. doi: 10.1016/S0140-6736(20)31792-X. Epub 2020 Aug 29. Erratum in: Lancet. 2020 Sep 12;396(10253):758.Ho CY, Olivotto I, Jacoby D, Lester SJ, Roe M, Wang A, Waldman CB, Zhang D, Sehnert AJ, Heitner SB. Study Design and Rationale of EXPLORER-HCM: Evaluation of Mavacamten in Adults With Symptomatic Obstructive Hypertrophic Cardiomyopathy. Circ Heart Fail. 2020 Jun;13(6):e006853. doi: 10.1161/CIRCHEARTFAILURE.120.006853. Epub 2020 Jun 5.
- Savsin H, Tokarek T. Comprehensive Review: Mavacamten and Aficmten in Hypertrophic Cardiomyopathy. Biomedicines 2025 Jul; 13(7):1619


 Translate
Translate